It was Albert Einstein who put forward the particle theory of light i.e Photoelectric Effect (hypothesized By Max Planck) that later paved the way for the creation of the Quantum World.
In the early 20th Century, a phenomenon like Interference, Diffraction, and Polarisation could only be demonstrated by assuming that Light Had A Wave Nature. But there were some other physical phenomena such as Blackbody Radiation, Photoelectric Effect, Hydrogen Spectrum, and Compton Scattering that could not be understood. The problems within Classical Physics led to the development of Quantum Theory.
Inadequacy of Classical Mechanics
1. Blackbody Radiation
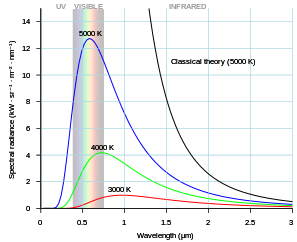 |
| Black Curve (Predicted By Classical Theory) Blue Curve (Predicted By Planck’s Theory) CREDIT: WIKIPEDIA |
The classical theory, predicts that “a hot body should radiate an infinite amount of energy directly contradicting the experimental evidence shown by some physicists as well as violating the principle of conservation of energy“.
The electrons in the hot object can vibrate with a different range of frequencies, from very few vibrations per second to a huge number of vibrations per second. Classical Physics predicts that each frequency of vibration should have the same amount of energy directly contradicting and indicating that a new model for the explanation of the blackbody spectrum was in need to be discovered.
In 1900, Planck suggested that each wave has a natural energy that is totally independent of the temperature of a hot body. Energy is not distributed equally by electrons that vibrate at different frequencies.
i.e
THAT IS WHY THE BLACKBODY SPECTRUM ALWAYS BECOMES SMALL ON THE LEFT-HAND (HIGH FREQUENCY) SIDE.
2. Photoelectric Effect
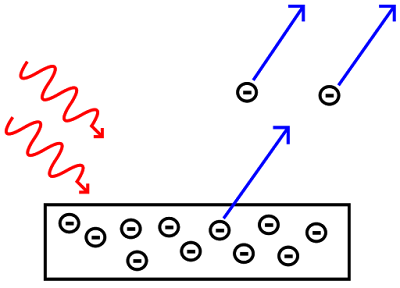 |
| Electrons emitted from metal plate via the photoelectric effect CREDIT: Wikipedia |
Nowadays photoelectric effect is used to produce the electric current that runs many solar-powered devices.
According to Classical Electromagnetic Theory or Wave Theory, light is a wave having energy distributed throughout the wave. Basically, classical physics made two hypotheses about the photoelectric effect;
1) A little adjustment in the intensity of light will change the kinetic energy of electrons emitted from the metal surface.
2) A sufficiently dim light will take some time for enough light energy to raise up and eject an electron from the metallic surface.
However, both of the above hypothesis was WRONG!
The experimental results did not match the two predictions made by Classical physics. It would make no difference how dim the light is. There would never be a time delay. The measurements were quite easy for Einstein to explain if the light is made up of particles (photons) instead of waves.
In 1905, Albert Einstein polished and extended the hypothesis made by Max Planck used to explain the blackbody spectrum that light must consist of clumps or quanta of energy later called photons. He assumed that each quantum of light possesses the energy hf (Planck’s constant times the frequency of the light).
The absorption of a single photon by an electron increases the energy of electrons by hf. Some part of this energy is used to eject the photoelectrons from the metal surface. This is called the Work Function. And the remaining energy is transferred to the electrons to increase their kinetic energy.
Therefore the energy of light is not evenly distributed along with the wave but is concentrated in the photons.
According to Einstein, a dimmer light means fewer photons, but simply turning down the light (without changing its frequency) does not alter the energy of an individual photon. So for a specific frequency light, if a single photon has enough energy to eject an electron from a metallic surface, then electrons will always be ejected immediately after the light is turned on and the photons hit the metal.
Einstein won his Nobel prize for showing that if a light is considered as a particle instead of a wave, whose energy is proportional to the associated frequency, then the photoelectric effect is fully explained.
3. Compton Effect
When the high-energy photon collides with the target in which electrons are loosely bonded to the nucleus, therefore, can be treated as free. As a result, the scattered radiation is found to have smaller radiation than the incident radiation. This is known as the Compton Effect or Compton Scattering.
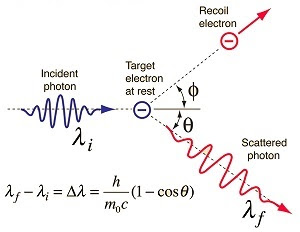 |
| Credit: hyperphysics.phy-astr.gsu.edu |
According to the classical view of Compton scattering or Classical Wave Theory, Electromagnetic Radiation scattered by charged particles cannot explain any type of shift in wavelength or frequency. Compton’s effect demonstrated that light cannot be explained purely as a wave phenomenon. In simple terms, the Compton Effect shows the particle nature of light
Compton scattering not only confirmed the law of conservation of energy which was verified by the Photoelectric Effect but also explained the law of conservation of Linear Momentum.
Although, it was Max Planck and Albert Einstein who hypothesized that light could behave as both a wave or particle. But, it was Arthur Compton who proved that this was possible.
4. Bohr’s Hydrogen Spectrum
In simple words, atoms should not survive longer than 10-12 seconds.
This clearly did not happen. Therefore, the prediction of classical theory came to be WRONG!
 |
| CREDIT: Quora |
Neil Bohr used Ernest Rutherford’s Model Of An Atom and the theory of quantum physics for describing the behavior of a model (Bohr’s Atomic Model) that was accepted for the better part of the 20th century.
Bohr postulated his theory much like Rutherford but placed charged particles (electrons) in layers of orbit and with each orbit having specific energy such that, electrons can jump from a higher orbit to the lower orbit, and when their energy decreases they return to their orbit.
In simple words, electrons have more energy when there are in larger orbits.
Bohr’s model’s key success was the explanation of the Rydberg formula for the spectral emission lines of the hydrogen atom. The quantization of atoms also contradicted the predictions made by classical theory.
At last, I will leave you all my readers with a quote by Neil Bohr regarding Quantum Mechanics, and he quote
“IF QUANTUM MECHANICS HASN’T PROFOUNDLY SHOCKED YOU, YOU HAVEN’T UNDERSTOOD IT YET. EVERYTHING WE CALL REAL IS MADE OF THINGS THAT CAN NOT BE REGARDED AS REAL”
That’s It For This Post. Feel free to comment and make me know if I have left something.
Please share It If You Like, Like It If You Share.
∗ Don’t Believe It As I Said Till You Yourself Believe It First – Gautama Buddha

I read your post and got it quite informative. I couldn't find any knowledge on this matter prior to. I am thankful to you for sharing this article here. Teacherlookup is the best online platform where you can find A Level Physics Tutor Online.
We have sell some products of different custom boxes.it is very useful and very low price please visits this site thanks and please share this post with your friends. supernatural stories