What Is Wave-Particle Duality?
Is light a wave or particle? Well, whether light is a wave or light is a particle has a very interesting and long history. Wave-particle duality or the Dual nature of light is the sole idea for the definition of the physical reality of quantum physics.
In other words, the duality of nature (wave-particle duality) stands for all the matters and light that exhibit the behavior of both waves as well as particles.
According to the laws of quantum mechanics, both light and matter can be described not only in terms of particle nature but also in terms of wave nature, of course; depending upon the circumstances of the wave-particle duality experiment.
Although, it is a complicated topic, yet among the most fascinating in the world of Physical Science.
Wave Particle Duality – Quantum Theory Of Light
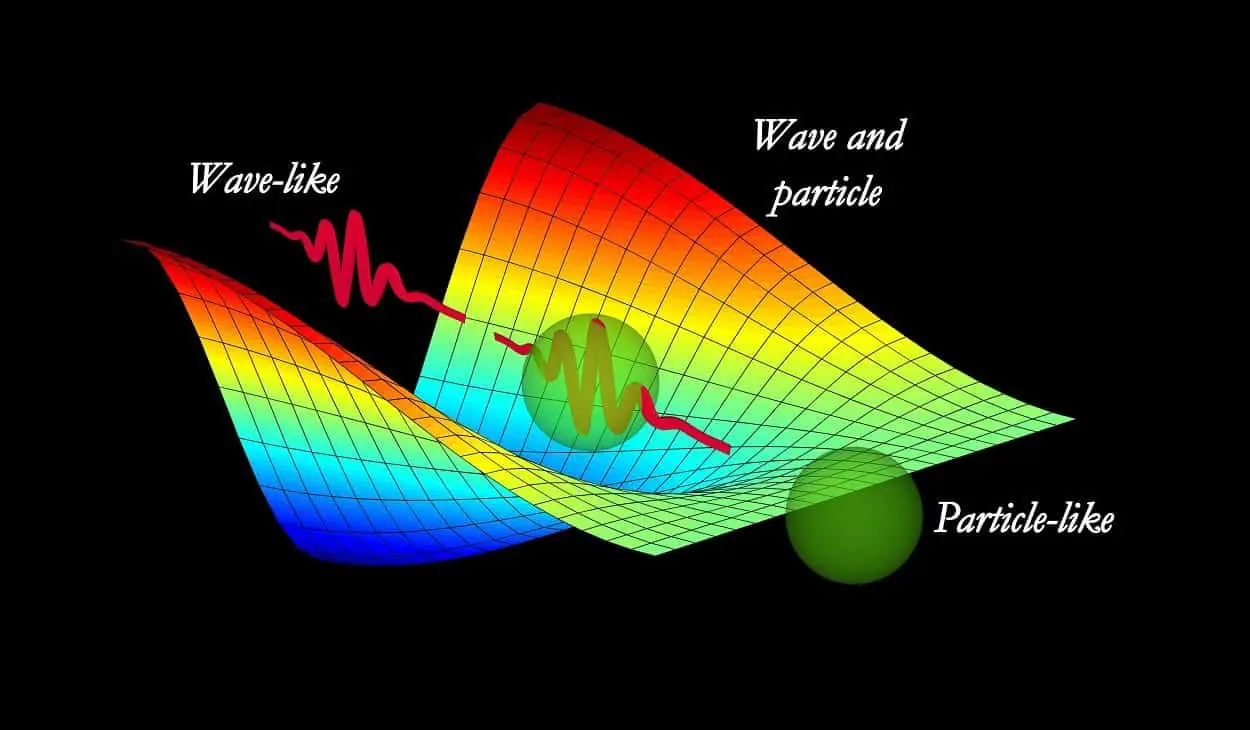
The conflict between the wave-particle duality of light i.e light is a wave or light is a particle, emerged centuries ago. As we discuss quantum theory, let us take a Quantum leap (but back in time).
As per records, Democritus was the first one to talk about the nature of light. In fact, he advocated that whatever there is in this universe is composed of an indivisible sub-component (particle nature of light).
Newton says Light Is A Particle
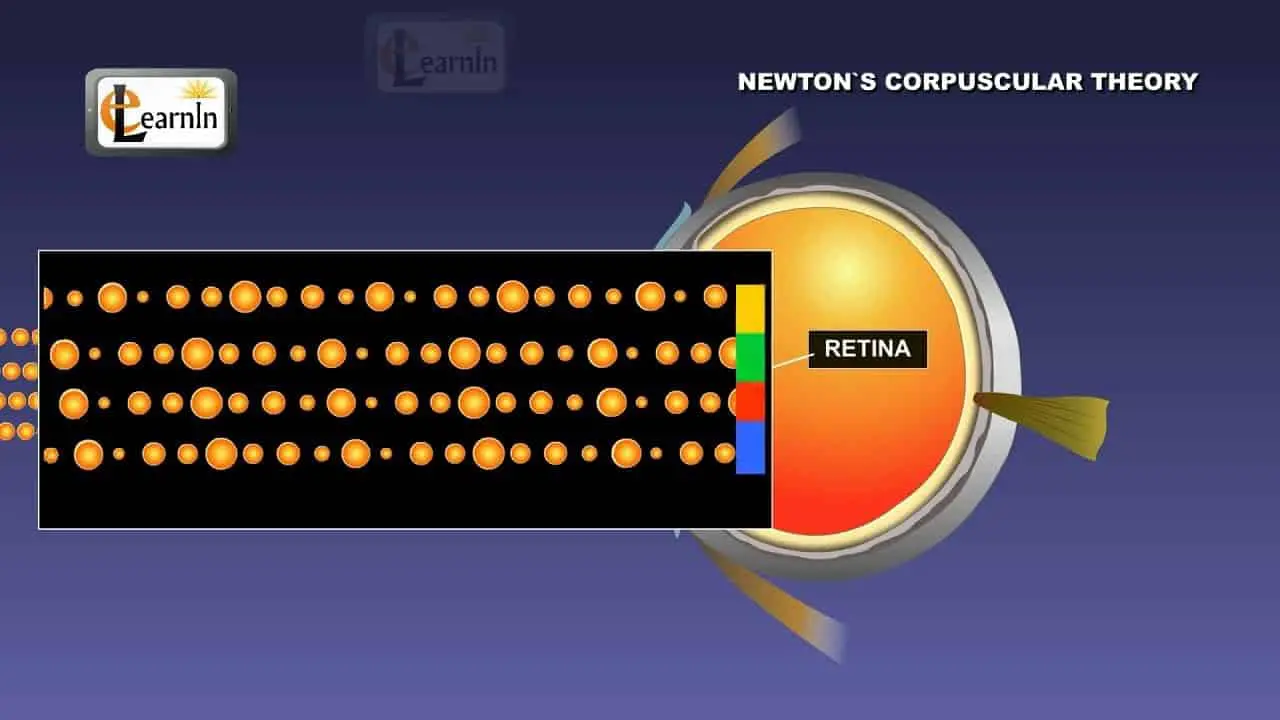
In the late 1600s, Issac Newton, “the greatest among the great” developed and championed his Corpuscular theory. Issac Newton believed that light was made of a collection of particles.
He proposed that a light source emits tiny corpuscles of light. Therefore, only particles could travel in such straight lines.
Not to mention, light appears to travel in a straight line and cast shadows behind the obstacles. Thus, it was conceivably the strongest evidence that light is a particle.
Huygens says Light Is Wave
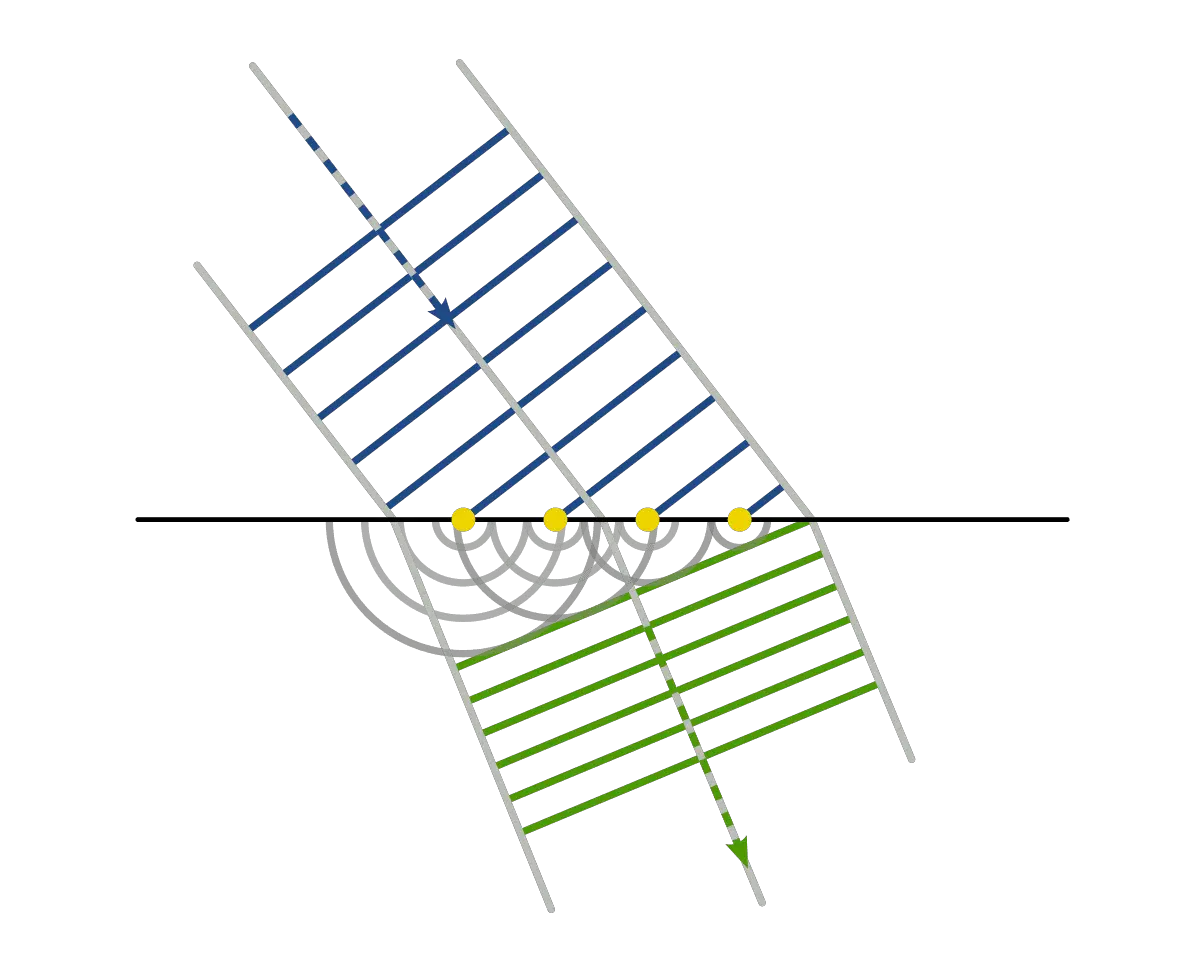
On the other hand, Newton’s greatest contemporaries, Christiaan Huygens suggested that light is wave.
Christian Huygens and his supporters for the Huygens wave theory of light showed that if light can travel at a different speed in different mediums (water, air, vacuum, etc). Therefore, refraction could be easily demonstrated.
According to the definition of Huygens principle, “the apparent rectilinear propagation of light may be due to the fact that the wavelength of light may be much smaller than the dimensions of the openings or obstacles”.
No, Light Is A Particle
On the contrary, Huygens Fresnel’s principle had some issues in matching observation. In other words, it could not explain the divergence from rectilinear propagation that appears when the light run-in into the edges, screen, or aperture.
An effect is collectively known as the Diffraction Effect. Therefore, as a result; the Huygens wave theory was discarded for almost a century.
Newton’s reputation and the belief of the scientific world in Newton’s writing held the Corpuscular Theory Of Light dominant for almost for a long period of time until the emergence of Thomas Young’s 1803 discovery of Double Slit Interference.
No, Light Is Wave

In the early Nineteen Century, complications arose for Newton’s Corpuscular Theory Of Light. The diffraction effect, which Huygens wave theory failed to explain was perfectly explained by Young’s Double Slit Experiment.
Thomas young experiment clearly certified that “light coming from the two coherent sources interferes and produces maxima and minima depending on the path difference”.
In other words, the Double Slit Experiment gave evidence that light is a wave. Therefore, resulting in the triumph of wave theory over particle theory of light”.
Light Does Not Need Medium To Propagate
In wave motion, there is some quantity that changes its value with respect to time and space. Likewise, in the case of the sound wave, it is the pressure at a point that oscillates as time passes.
On the other hand, in the case of light, the light wave is quite different from the rest. Light is a non-mechanical wave. Lightwave doesn’t need a material medium to propagate. They can travel in a vacuum.
Though, earlier, physicists used to assume that a very dilute and highly elastic medium is present everywhere in space, named “Aether”.
Recommended, Aristotelian Universe – Let’s Go Back In Time
In fact, throughout the nineteenth century, various attempts were made to detect Aether. However, the Michelson Morley Experiment confirmed the absence of Aether.
Aether was never discovered to date. For this reason, we assume or understand that light waves can propagate in a vacuum only. Consequently, a huge debate as the twentieth century began. Is Light A Wave Or Particle? After all this strange happening, the drama was not over yet…!!!
No, No, Light Is A Particle
The climax came when the classical wave theory was unable to explain the Philipp Lenard experiment.
Lenard observed that “when light rays fall on the metal surface, electrons are ejected and the kinetic energy of the emitted electrons does not depend on the intensity of the light used”.
Take a look at EPR Paradox, a Thought, Myth, or, a Reality?
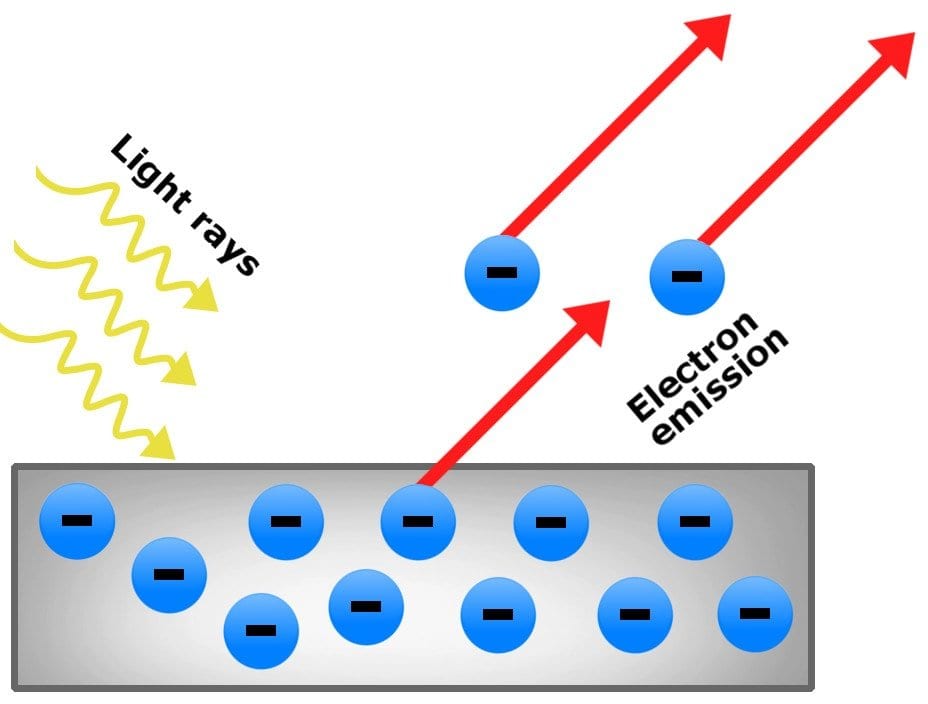
Though, Heinrich Hertz had the first observation of this phenomenon in 1887. This phenomenon is known as the Photoelectric Effect.
Later, another giant, Albert Einstein, explained the Photoelectric Effect Experiment in his 1905 research papers. Einstein proposed that the energy contained within a photon is related to the frequency of light.
Truce
The old question “whether light is wave or light is a particle” was reopened and harmonious understanding was reached in accepting that light has a dual characteristic. As Albert Einstein states:
It seems as though we must use sometimes the one theory (particle theory) and sometimes the other (wave theory). We are faced with a new kind of difficulty. We have two contradictory pictures of reality; separately neither of them fully explains the phenomena, but together they go.
Wave Particle Duality – Quantum Theory Of Matter
A young French physicist Louis Broglie, still in his twenties, suggested that if light can have a dual character i.e wave-particle duality of light. Then, why can’t materials like electrons can have the same characteristics i.e electron as a wave or particle?
In other words, he simply had a thought; what if particles like electrons and protons also behave like waves i.e dual nature of matter Therefore, he proposed that any object or a moving particle had an associated wave (matter waves).
The question of whether such dual nature (as shown by light) can also be exhibited for the matter was bravely tackled by the bold de Broglie Hypothesis.
De Broglie’s relation was simply based on the work of Max Planck and Albert Einstein. Broglie used Planck’s Quantum theory and Einstein’s photoelectric equation to narrate the observed wavelength of matter (de Broglie wavelength) to its momentum.
In De Broglie’s view, the sum of the total energy of this vast universe can only be explained in the form of Radiation and Matter.
Therefore, both radiation and matter should have similar properties. And accordingly, finally giving the expression which we now call as De Broglie Wavelength Equation.
where,
h = Planck’s Constant
p = momentum of a particle
λ = de Broglie wavelength
Confirmation Of Matter Waves
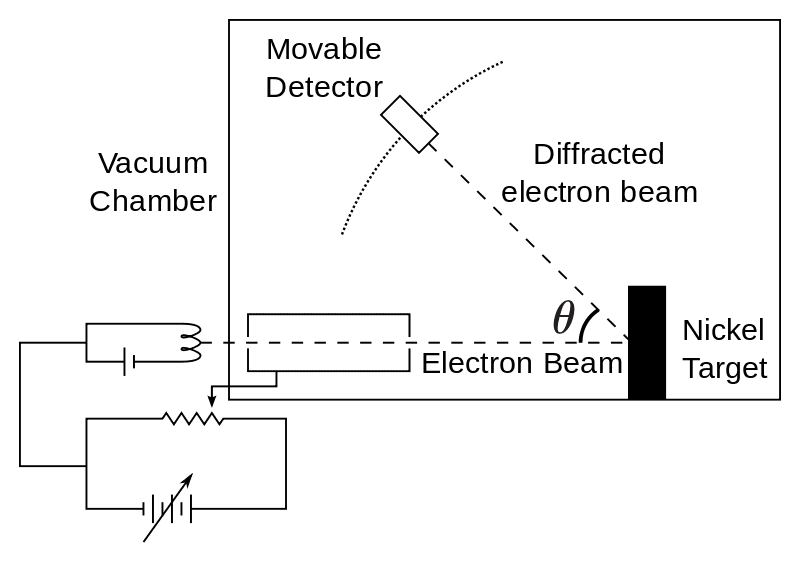
His work on matter waves was so bold that it gathered criticism from many pioneers of the scientific world such as Wolfgang Pauli etc. But, that was not the case with Albert Einstein. In fact, Einstein warmly supported the De Broglie conclusion.
Later in the coming years, his hypothesis for matter waves was confirmed by American physicists Clinton Davisson and Lester Germer in their experimental demonstration named Davisson-Germer experiment.
Therefore, resulting in a 1929 Nobel prize for physics for Louis de Broglie.
At last, I will leave you all my Physics In My View Readers with a quote by Richard P. Feynman regarding Quantum Theory, and he quotes:
If I say [electrons] behave like particles I give the wrong impression; also if I say they behave like waves. They behave in their own inimitable way, which technically could be called a quantum mechanical way. They behave in a way that is like nothing that you have seen before.
That’s it for this post. If you like this article, share it if you like it, like it if you share it. You can also find us on Mix, Twitter, Pinterest, and Facebook.


ATUL SINHA New innovation in Super chemical low energy nuclear reactions involving Wave Particle Duality Simon Brink http://www.subtleatomics.com Very interesting technology .
Frankly speaking, I haven’t read about it. But, I am looking forward to it.