Quantum Physics was progressively derived from Max Planck’s solution to the black-body radiation problem in 1900. Later it was extended by Albert Einstein’s 1905 paper which offered a Quantum-Based Theory to explain the Photoelectric Effect.
If you are reading something related to the theories of quantum mechanics, here is the list of Top 6 Quantum Physics Fun Facts You Can’t-Miss. With that, you will find Quantum Physics will become a lot easier to understand.
Top 6 Interesting Facts about Quantum Physics
If you can’t relate to the top 6 interesting and weird facts about quantum physics, well, here is your chance to think again…!!!
- Quantum Physics is Totally Discrete in Nature
- In Quantum World, Everything Depends on Chance
- Quantum Physics is Highly Dependent on Wave-Particle Duality
- Quantum Physics in Incomplete
- Even Stars Follow Quantum Laws
- Quantum World has Black Holes
Quantum Physics is Totaly Discrete in Nature

The very first one in my list of Top 6 cool and weird facts about quantum physics is its discreteness. I mean if Max Planck (father of quantum physics) hasn’t thought about the discrete nature of energy distribution (Quanta Of Energy), there could be no Quantum Theory.
In other words, the laws of quantum mechanics strictly follow the discrete nature of energy distribution. The energy accommodated in the Quantum Field Theory comes as integer multiples of some fundamental energy.
To put it differently, the discrete nature of quantization is a process of adaptation from the classical understanding (macroscopic) of physical phenomena to the modern understanding of physical phenomena (microscopic).
Moreover, the word quantum descended from the Latin word account for “how much” or “how great”. As it refers to a discrete value specified to some acknowledged physical quantities referred to as the energy of photons.
In Case of Light
For light, this discrete energy or quanta of energy is related to the frequency and wavelength characteristic of the light.
- High-frequency, short-wavelength has a large characteristic energy level.
- Low-frequency, long-wavelength has a small characteristic energy level.
Take a Note
Suppose, as in both cases, if energy levels were continuous, we would expect there should be an infinite amount of energy levels just as there are infinitely many fractions that come between one and two.
Since the energy levels are discrete, therefore, there are finite integer multiples of energy levels such as 1, 2, 94, 167, etc; never a weird fraction like π, three-and-a-half, or the square root of two.
Otherwise, it would directly come on the list of Most Bizarre Inadequacies Of Classical Physics. In fact, the meaning of quantum physics or the word quantum itself means the smallest distance between the two different discrete energy levels.
On the other hand, if I talk about the applications of quantum mechanics, of course, depending upon its discrete nature, then Solid-state physics, condensed matter state, ultra-precise spectroscopy, and, even Atomic clocks work because of the discreteness of quantum physics.
In Quantum World, Everything Depends on Chance

Whenever any quantum physicist introduces quantum mechanical theory to any system to predict the outcome of an experiment. For him, it is impossible to predict the certain outcome of a single experiment on a quantum system. In fact, it is one of the most amazingly disputable aspects of quantum mechanical theory that hasn’t been solved yet.
As a matter of fact, the only thing they can predict is the probability of detecting each of the discrete possible outcomes of a quantum system. Therefore, the comparison between theoretical predictions and experimental outcomes always requires inferring probability distribution from many repeated experiments.
Take an Example
Suppose an experiment in which a photon (quanta of energy) will end up in one of the two places. As a result, we can say that there is a 42% chance (probability) of finding it at a point named A.
On the other hand, there is a 58% chance (probability) of finding it at a point named B. But, we will never be sure that a single photon will surely end up at point A or at point B.
To put it another way, it doesn’t matter how carefully we measure the position of a photon, we can never be so sure of what the outcome of the experiment will be. The final outcome will always be random. Well, at least for now.
Quantum Physics is highly dependent on wave-particle duality
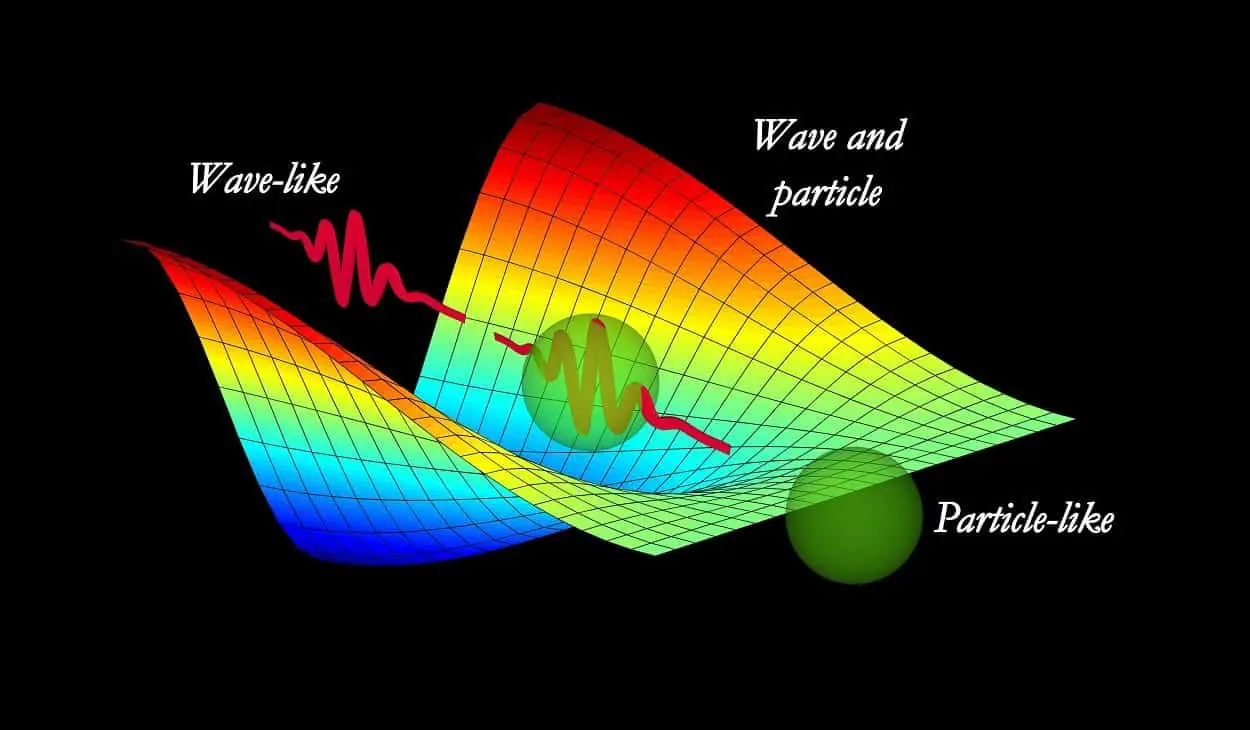
One of the most important facts about quantum physics is its love for the duality of nature. Of course, not that love you are thinking of right now.
Whatever there is, whatever there was, whatever there will be, can only be explained within the realm of the wave-particle duality of nature.
I mean, in quantum physics, if you want to explain any phenomenon, you have to explain it in terms of wave nature or particle nature. Or, maybe both.
Everything in the universe has both particle-like properties as well as wave-like properties at the same time. In other words, wave-particle duality comes in the realm of the basics of quantum physics.
Through the work of Albert Einstein, Max Planck, Louis de Broglie, Neil Bohr, Arthur Compton, and many others, our current quantum mechanical theory holds that all particles are waves (and vice versa).
At last, you can say that every object in the universe described by quantum physics is neither a particle nor a wave. In fact, It can be regarded as a new kind of object; call it a “quantum particle”.
A particle that exhibits some characteristics of waves (spread over space), as well as, some characteristics of particles (discrete and countable).
In fact, this wave-particle duality phenomenon has been confirmed not only for elementary particles but also for the compounds like atoms and even molecules.
Einstein View on Wave-Particle Duality
As once Albert Einstein quoted regarding wave-particle duality:
It seems as though we must use sometimes the one theory and sometimes the other, while at times we may use either. We are faced with a new kind of difficulty. We have two contradictory pictures of reality; separately neither of them fully explains the phenomena of light, but together they do.
Quantum Physics in Incomplete

One of the most unusual and bizarre outcomes of Quantum Physics was the idea of “Quantum Entanglement”.
In 1935, Albert Einstein and his younger associates Boris Podolsky and Nathan Rosen collectively published an outstanding paper (EPR Paradox) which was to become the last great contribution of Einstein to mankind.
What is EPR Anyway?
Well, Einstein Podolsky Rosen paradox, or simply the EPR paradox is an influential thought experiment regarding the idea of quantum mechanics interpretation.
If you don’t know, the currently accepted quantum mechanics interpretation is the Copenhagen interpretation of quantum mechanics. Copenhagen interpretation is the brainchild of Neil Bohr and his esteemed colleagues.
Apart from the Copenhagen interpretation, there are so many other quantum interpretations that I don’t wanna discuss here. Maybe, I would write a proper article especially discussing the different interpretations of quantum mechanics.
Simply, because this quantum interpretations topic is too deep to be discussed here. As once, quantum physicist N. David Mermin said, “New quantum interpretations appear every year, None ever disappear”. I hope, now you realize the extent of this topic.
Spooky Action at a Distance
According to the EPR paradox, quantum particles can interact in such a way that it is possible to measure the position and momentum of particles more accurately than ever.
In other words, if the EPR paradox is correct, then this implies that information is being transferred at a speed faster than the speed of light, an idea that we now call “Quantum Entanglement”.
On the contrary, this controversial paper was directly contradicting the Heisenberg uncertainty principle and Einstein’s own theory of relativity.
In reality, even Einstien knew the consequence of the entanglement theory. That’s why he described it as “spooky action at a distance”.
Take an Example
When two quantum particles interact in a conventional way, their states will depend on one another. It doesn’t matter how far apart they are.
Suppose a quantum particle is in India and another one is in Alaska. When you measure them simultaneously, the outcome of the measurement of a quantum particle in India will unquestionably and undisputably determine the outcome of the measurement of the other quantum particle in Alaska and vice versa.
In Einstein Podolsky Rosen’s view, the wave function does not provide a complete description of physical reality. Hence, the EPR paradox showed that quantum theory is indeed incomplete. Therefore, should be extended with some local hidden variables.
Einstein was Wrong

In the year, 1963, Irish quantum physicist John Stewart Bell proved that quantum mechanical theory is incompatible with the local hidden variable theories; one that was proposed by Einstein in his EPR argument.
According to Bell’s Theorem, the only way hidden variables could predict the outcomes of the quantum mechanical world, only and only if hidden variables are non-local.
In fact, as Bell summarises his view about the EPR argument, he quotes:
If a hidden variable theory is local, it will not agree with quantum mechanics; and, if it agrees with quantum mechanics it will not be local
Take Away Points
As you can see there are two conflicting statements regarding the laws of quantum mechanics, therefore, directly confronting each other.
In fact, both arguments agree with the fact that quantum entanglement is real. On the contrary, they debated the fact that whether the quantum mechanical theory is incomplete or not.
- In Einstein’s view, quantum mechanics is incomplete, therefore, needs local hidden variables to complete it.
- In Bell’s view, quantum mechanics is complete, therefore, local hidden variables are incompatible with the laws of quantum mechanics.
Well, as per experimental pieces of evidence are concerned, I am afraid to say that Einstein was wrong. Quantum physics is complete. But yes, you can say that quantum physics is non-local.
Meaning, that maybe the EPR argument was proven wrong, but, indeed, we got entanglement theory out of it; which is actually quite real.
On the other hand, if I talk about the applications of quantum mechanics, of course, depending upon its non-locality, Quantum entanglement has applications in the emerging technologies of quantum computing and quantum cryptography.
In fact, it has been used to accomplish quantum teleportation experimentally. Not to mention, there are some quantum physicists who are trying to find some correlation between entangled particles and wormholes.
Well, who knows, what could be possible in the near coming future. I mean, it took almost a century for the prediction of black holes to become reality. But, indeed it has become a reality.
Even Stars Follow Quantum Laws
Yup, It’s true. The next one on my list of top 6 quantum physics facts is that even stars follow quantum laws. Well, it’s not a hidden fact that stellar fusion or nuclear fusion empowers the life of a star for most of its existence.
On the other hand, Nuclear reactions are a byproduct of Einstein’s mass-energy equivalence that is e=mc2, which is nothing but a quantum mechanical phenomenon.
Therefore, by all this correlation, I can for sure say that in order to shine, even Stars follow quantum laws. As a matter of fact, both star’s life and the death of a star are somehow related to the laws of quantum mechanics.
Editor’s Choice: Why my Top 6 Nuclear Energy Facts are BETTER than Yours?
Death of a Star

First of all, don’t worry. I mean, I won’t bother you with some harsh astrophysical words. I will tell you everything thing in layman.
A star is nothing but a ball of hydrogen gases. See, as you know that stars shine because of nuclear fusion. On the other hand, e=mc2 strictly states that energy and mass are equivalent to each other.
Meaning, that in order to shine, the mass of a star (fuel) must be utilized. Therefore, when the stellar core of a star runs out of its nuclear fuel, BOOM, it collapses under its own gravity. Hence, a star dies. Again, here comes the role of the laws of quantum mechanics.
Electron Degeneracy Pressure

First of all, the stellar death of a star doesn’t mean the end of the story of a star. On the contrary, it has just begun. This means that a dying star can either transform itself into a white dwarf, or a neutron star. Or, a dying star could even become a black hole, of course, depending upon its stellar mass.
Therefore, when a star collapses under its own weight, it’s the electron degeneracy pressure that comes into effect and, protects a dying star from further collapsing.
This means that when electron degeneracy pressure counterbalances the effect of gravity, a white dwarf is born. Well, of course, there is some mass limit. After the stellar death of a star, the mass of the remnant core of a star must be smaller than or equal to 1.34 solar masses.
This mass limiting formula for the formation of a white dwarf is known as the Chandrasekhar limit. On the other hand, if I talk about the electron degeneracy pressure, it’s nothing but a manifestation of quantum degeneracy pressure, which is also perceived as one of the basics of quantum physics.
Neutron Degeneracy Pressure

In a similar fashion, when the remnant mass of the stellar core of a star exceeds the Chandrasekhar limit, the neutron degeneracy pressure will come into effect. Therefore, protects the dying star from further collapsing to become a black hole. Hence, a neutron star is born.
Again, just like the Chandrasekhar limit, there is some mass limit for the formation of neutron stars. Meaning, that after the stellar death of a star, the mass of the remnant core of a star must be greater than 1.34 solar masses, but, between 2 or 3 solar masses.
In fact, if the remnant star exceeds that limit, it will eventually meet its final fate i.e a black hole. This mass limiting formula for the formation of neutron stars is known as the Tolman Oppenheimer Volkoff limit or TOV limit.
Quantum World has Black Holes
Well, as far as theoretical predictions are concerned, just like the relativistic world has black holes, In a similar fashion, the quantum world too has black holes. Quantum black holes are classified as micro black holes, mini black holes, or simply miniature black holes.

In fact, whenever we talk about the different types of black holes, quantum black holes are included in that list. Apart from quantum black holes, the other types of black holes are stellar black holes, intermediate-mass black holes, supermassive black holes, and of course, primordial black holes.
Related, Wait, WHAT? Strange Metals Are Like Primordial Black Holes?
Not to mention, Quantum black holes haven’t been found yet. They are still theoretical predictions to date. Well, who cares. I mean, the first theoretical predictions of black holes were done by einstein in 1915.
Yet, the first-ever black hole was discovered in 2019, a century after its predictions. Therefore, maybe in the coming decades or so, we could even discover some quantum black holes.
What If?
Recently, an international research collaboration has announced the witness of the birth of an “intermediate-mass” black hole. Before this discovery, they were thought to be hypothetical ones, just like quantum black holes.
What I mean to say, is if an intermediate-mass black hole can be found, then why not the quantum black holes. Well, what’s your take on this? How much time astronomers and researchers would take to finally discover a unique quantum black hole?
That’s it for this post. If you like this article, share it if you like it, like it if you share it. You can also find us on Mix, Twitter, Pinterest, and Facebook.
