Wanna know what are some unveiling examples of molecules in daily life? If yes, then you are at the right place at the very right time. Because in this exclusive article, I am gonna explain how molecules play a significant role that impacts our everyday life experience. So, what are molecules anyway?
When two or more atoms combine due to chemical bonds with each other, what we get is a molecule. We cannot see them with the naked eye. However, molecules are visible through a magnifying microscope.
Understanding the concept of molecules that whether it is made of the same atom or different from each other can help us comprehend the world around us in a much better way. So, without wasting any more time, let’s dive right in…!!!
Examples of Molecules Made Simple: A Quick Reference Guide
- Chlorophyll
- Asprin
- Glucose
- Ethanol
- Caffeine
- Water
Chlorophyll

The very first one on my list of exclusive examples of molecules in real life is chlorophyll. It is a molecule responsible for the green pigment in plants and algae. The molecular structure of chlorophyll consists of a chlorin ring, whose four nitrogen atoms surround a central magnesium atom, and has several other attached side chains and a hydrocarbon tail formed by a phytol ester.
Apart from being responsible for the green color of plants, it also plays a crucial role in the process by which plants convert sunlight into energy. Chlorophyll absorbs sunlight and facilitates the chemical reaction to convert carbon dioxide, and water into glucose, and food and release oxygen, hence, sustaining life on Earth. The molecular formula for chlorophyll is (C₅₅H₇₂O₅N₄Mg).
Asprin
Acetylsalicylic acid, commonly known as Aspirin, is a molecule used as an analgesic, antipyretic, and anti-inflammatory drug. It is widely used to alleviate pain, reduce fever, and mitigate inflammation. The molecular formula for Aspirin is (C9H8O4).
Its molecular structure allows it to interact with specific enzymes and receptors in our bodies. Therefore, as a result, provides pain relief, and reduces inflammation. Generally, Aspirin is used to alleviate headaches, reduce fever, and prevent cardiovascular events.
Glucose
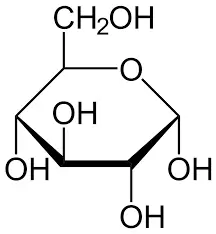
Glucose is a carbohydrate molecule that serves as the primary source of energy in our bodies. Living organisms, ranging from bacteria to plants and animals, use glucose as a vital energy source. It is basically found naturally in food and is derived from the breakdown of various foods and fuels we consume in our day-to-day life.
Without glucose, our body won’t survive for long. In fact, it is vital for powering our cells and ensuring the proper functioning of our bodily organs. The molecular formula for glucose is (C6H12O6). In addition, it has a sweet taste which is why glucose is commonly known as a SUGAR.
Ethanol

Ethanol or also known as ethyl alcohol is a molecule found in alcoholic beverages. Apart from being used as alcoholic beverages, it has a wide range of practical applications in our daily life. Such that it is most commonly used as a solvent or fuel source in our various industries.
In fact, Ethanol is also used in the healthcare industry as a disinfectant and antiseptic. It is colorless and produced through the process of fermentation of sugars found in fruits, grains, or other plant material. In addition, the molecular formula for Ethanol is (C2H5OH).
Caffeine

Caffeine is a natural stimulant that belongs to the class of compounds known as methylxanthines. The molecules of caffeine are found in coffee beans, tea leaves, cocoa beans, and kola nuts. Since it directly acts as a central nervous system stimulant, it can have both positive and negative effects on the human body.
One of the primary effects of the use of caffeine is that it increases alertness and reduces fatigue. The scientific data available suggests that it basically blocks the action of adenosine, a neurotransmitter that promotes relaxation and sleepiness. The molecular formula for caffeine is (C8H10N4O2).
Water

Last but not least one on my list of various examples of molecules is water, the elixir of life. Water’s unique properties, such as its high heat capacity, surface tension, being a solvent, etc, make it indispensable for human existence. In other words, we use water in almost every aspect of our life.
Be it for drinking purposes, cooking, cleaning, or even washing, water is used everywhere. In fact, its ability to exist in solid, liquid, and gaseous states, makes it more relevant than any other substance available on Earth. The molecular formula for water is (H2O).
Some Other Examples of Molecules in Daily Life
Apart from the above-mentioned ones, I am also mentioning a few here.
- Oxygen
- Carbon dioxide
- DNA
- Sodium Chloride
- Sucrose
- Cholesterol
- Dopamine
- Serotonin
- Melatonin, etc.
That’s it for this post. If you like this article, share it if you like, like it if you share it. You can also find us on Mix, Twitter, Pinterest, and Facebook. Hey man, If you have come this far, do give us feedback in the comment section. It would make my day. You can also make a donation. Your donations will help us to run our website and serve you BETTER. Cheers!!!
You might also like:
- Anion vs Cation – What’s the Difference??
- Ionic and Covalent Bonds – Difference & Similarities
- Common Covalent Compounds in Everyday Life – Top 6
- Examples of Chemical Energy in Everyday Life – Top 6
- Exothermic Reaction Examples in Everyday Life (Exclusive)
- 10 Examples of Endothermic Reactions in Everyday Life
